|
8/12/2023 0 Comments Co element or comoundThere is some empirical and theoretical evidence for a few metastable helium compounds which may exist at very low temperatures or extreme pressures.Lesson 3: Names and formulas of ionic compounds. The ions, Ne+, +, +, and + are known from optical and mass spectrometric studies. Preferred names in the nomenclature of organic compounds: Draft 7 October 2004 IUPAC, 2004. Extension and Revision of the Nomenclature for Spiro Compounds (Including Bicyclic Compounds) (IUPAC Recommendations 1999). CH3CHCCH3 O CH3 C-N-CH3 Question Name the following from carboxylic acids, esters, amides, and amines Transcribed Image Text: 1.Moss, G. Name the following compounds using IUPAC nomenclature. CH 4 Correct Answer: Option C ExplanationName the following compounds using IUPAC nomenclature. The following rules enable us to identify any organic compound with a unique name:Which of the following molecules has a linear structure? A. The names of organic molecules are derived from the simplest of organic families. Which of the following structures contains secondary amine? NH2 NH NH2 IV A.I B.II C. When the temperature reaches the boiling point of any substance in the mixture, it will boil and can be collected as a pure gas, leaving the other components still in the liquid state.Name the following moleculesWhich of the following correctly matches the molecules to the names of the functional group? I. Air can be separated into the component gases in the mixture by cooling it until it liquifies, the slowly raising the temperature of the liquid air. Molecules of carbon dioxide are formed from one carbon atom and two oxygen atoms chemically bonded together. Argon is made of individual argon atoms not joined to anything else. Molecules of oxygen are made from pairs of oxygen atoms chemically bonded together. It contains elements such as argon and oxygen, and compounds such as carbon dioxide. There are many physical means of separating a mixture: the right one to use depends on the physical states of the substances in the mixture. Unlike a compound, the different substances in a mixture can be separated from one another without breaking any chemical bonds. 
The substances in a mixture can be elements, compounds, or both. MixtureĪ mixture contains more than one chemical substance, and therefore is not pure. It is not possible to separate the atoms in a compound without breaking chemical bonds. 
All pure substances are either elements or compounds. CompoundĪ compound is a pure chemical substance, but it is made from more than one type of atom, chemically bonded together. The periodic table collects and organises all the known types of atom from which the elements are formed. For example, the element chlorine consists of pairs of chlorine atoms chemically bonded together, so its chemical formula is Cl 2. 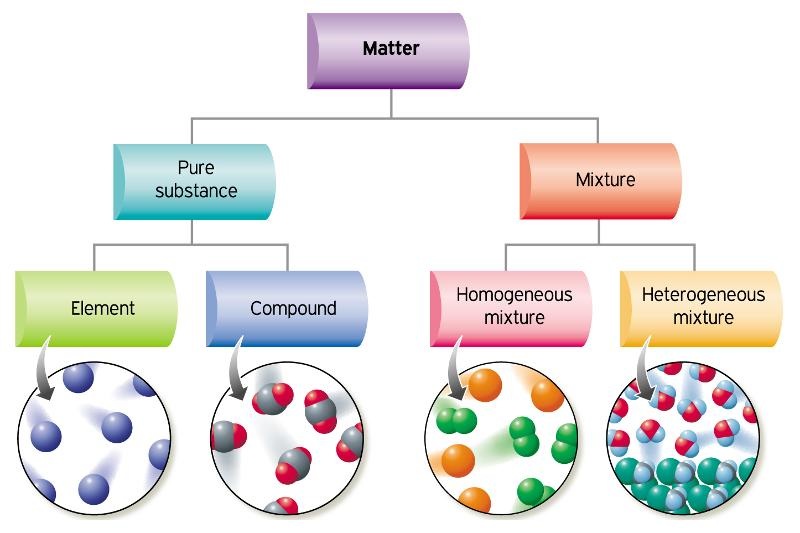
They may be joined to one another by chemical bonds, forming identical small groups of atoms called molecules. These atoms can be individual and separate, like in Noble Gases such as helium. 
ElementĪn element is a pure substance made up of only one type of atom. You should understand the difference between an atom and a molecule. Prior learning: You should understand that everything is made of atoms, and understand the differences between different types of atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
